This is a joint FSA and FSS publication.
1. Introduction
In accordance with assimilated Regulation 2015/2283 on novel foods, the application RP2124 for the use of tetrahydrocurcuminoids (THCus) has been submitted for authorisation in each nation of Great Britain (GB).
Whilst it was a Member State of the EU, the UK accepted the risk assessments of the European Food Safety Authority (EFSA) in respect of authorisations for regulated food and feed products. When GB left the EU, it retained the same regulations for food and feed regulated products; the FSA and FSS also adopted equivalent technical guidance and quality assurance processes (EFSA NDA, 2016) to be able to undertake GB risk assessments for regulated product applications.
To ensure our regulatory systems are risk proportionate and resources are used effectively, FSA and FSS have used the evidence submitted by the applicant and other information in the public domain, including the EFSA risk assessment opinion, to inform this safety assessment.
The FSA and FSS reviewers have evaluated the published EFSA risk assessment on the novel food and confirmed that this is appropriate for GB risk analysis. Consideration has been given to the processes undertaken to ensure the EFSA opinion is robust and whether there are any aspects that would require further review, such as specific issues for the countries of GB. The result of the assessment is that there is sufficient evidence of safety to conclude without requiring further risk assessment at this time.
This assessment represents the opinion of the FSA and FSS.
2. Details of other Regulators Opinions
The applicant, Sabinsa Europe GmbH, is seeking authorisation for tetrahydrocurcuminoids (THCus) as a novel food for use in food supplements, produced by hydrogenation of curcuminoids from turmeric (Curcuma longa L.).
In October 2021, EFSA published a risk assessment opinion on the safety of THCus for its use as a novel food, and it received a positive opinion (EFSA NDA Panel, 2021). This opinion has been reviewed by the FSA and FSS risk assessors.
THCus are also Generally Recognised As Safe (GRAS) in the USA.
2.1. Methodology applied in the EFSA Opinion
The EFSA Assessment was completed in accordance with the procedure as outlined in the EFSA scientific opinion ‘Guidance on the preparation and presentation of an application for authorisation of a novel food in the context of Regulation EU 2015/2283’ and Commission Implementing Regulation 2017/2469.
2.1.1. Identity of the novel food
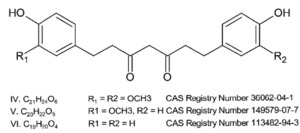
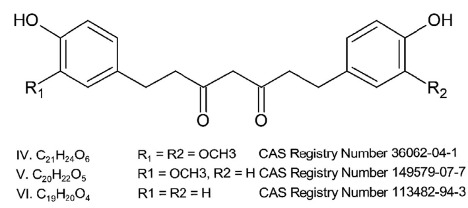
The novel food is a purified creamy white to pale yellow crystalline powder containing > 95% THCus by dry weight. The novel food is obtained by hydrogenating curcuminoids from the rhizomes of the turmeric plant, Curcuma longa. Three hydrogenated curcuminoids compose the novel food: tetrahydrocurcumin for curcumin, tetrahydrodesmethoxycurcumin for monodesmethoxycurcumin and tetrahydrobisdemethoxycurcumin for bisdemethoxycurcumin, respectively (Figure 1).
The main derivative is tetrahydrocurcumin (average percentage 87%), with tetrahydrodesmethoxycurcumin (average percentage 6.4%) and tetrahydrobisdemethoxycurcumin (average percentage 0.3%) present in lower amounts.
The identity of the three THCus composing the novel food was confirmed using high performance liquid chromatography with UV detector (HPLC-UV) at 280 nm, in addition to 1H- and 13C- Nuclear Magnetic Resonance (NMR) spectroscopy.
The FSA and FSS agreed with EFSA that the identity of the novel food is appropriately characterised.
2.1.2. Production Process
The novel food is manufactured using a two-step extraction and hydrogenation process. The raw material for the novel food, the rhizomes of Curcuma longa, are cleaned with water, steamed, and sun dried. Dried rhizomes are then pulverised and pelletised before the curcuminoids are extracted and dried to form crystals. The crystals are hydrogenated into THCus using palladium on carbon (Pd/C) as a catalyst.
As recognized by EFSA, the first part of the process corresponds to the traditional process used for the production of the food additive curcumin E100 (EFSA ANS Panel, 2010).
The novel food is produced in accordance with GMP and ISO 22000:2005 for the manufacture of standardised herbal extracts, phytonutrients, minerals and speciality fine chemicals and in accordance with ISO/IEC 17025:2005 for testing and calibration laboratories.
EFSA concluded that the data provided on the production process was sufficient, and as such did not give rise to any safety concerns or hazards for management. Upon review, the FSA and FSS agree with this decision.
2.1.3. Compositional information
The novel food is produced by catalytic conversion of curcuminoids, and consists of more than 95% tetrahydrocurcuminoids. Five independent batches of the THCus were characterised using high performance liquid chromatography with a UV detector, and demonstrated the ratio of the different THCus within the NF.
The full specifications for components of THCus provided by the applicant for 10 independent batches of the NF are shown in Table 2. The components of all were within safe ranges with minimal variation, demonstrating that the novel food can be produced consistently.
The compositional data on residual solvents (ethyl acetate, acetone, ethanol and isopropanol) and heavy metals (lead, cadmium and mercury) were found to comply with regulatory limits, as set by The Food Additives, Flavourings, Enzymes and Extraction Solvents (England) Regulations, 2013 and assimilated Regulation (EC) No 1881/2006 respectively (European Commission, 2006).
Microbial analysis of the NF was also performed and did not raise safety concerns (total aerobic microbial count, total yeast/moulds count, E. coli, Salmonella, S. aureus, P. aeruginosa, Bile-tolerant Gram-negative bacteria, Coliforms).
The presence of mycotoxins for five batches of the NF (aflatoxins B1, B2, G1, M1, ochratoxin A, zearalenone and fumonisin B1 and B2) were analysed and found to be below regulatory limits in food as set by assimilated Regulation (EC) No 1881/2006 (European Commission, 2006).
The FSA and FSS agreed with the EFSA assessment that the information on the composition is sufficient for characterising the novel food.
The stability of three batches were tested for 60 months at 30°C and 65% relative humidity and under accelerated conditions (6 months at 40°C and 75% relative humidity). No changes were observed to the content of THCus or the microbiological profile for the NF, indicating the novel food is stable under the conditions tested.
The applicant provided sufficient stability data to confirm a shelf-life for the novel food of 60 months from the date of production when stored under ambient conditions.
The FSA and FSS agreed with the EFSA assessment that the information on the stability of the novel food does not raise a concern on stability.
The FSA and FSS agreed with the EFSA assessment that the information on the stability of the novel food does not raise concerns on stability for further review.
2.1.4. Specification
Specifications of the novel food were provided by the applicant to detail the attributes and characteristics of the product. Five independent batches of the THCus were characterised using high performance liquid chromatography with a UV detector, and demonstrated the ratio of the different THCus within the NF, with more than 95% of the novel food consisting of tetrahydrocurcuminoids, demonstrating that the novel food can be produced consistently.
Certification was provided to demonstrate that the contract laboratories were accredited to perform these analytical studies. Where in-house analysis was utilised, full methodology and supporting validation documentation was provided. The data presented indicate the novel food can be consistently produced within the proposed specification.
The FSA and FSS agreed with the EFSA assessment that the information on the specification is sufficient to appropriately characterise the novel food.
2.1.5. History of Use
There is no history of use of the novel food in Europe or UK prior to 2019. An EFSA assessment for the same novel food achieved a positive opinion for the same intended food categories (European Commission, 2022).
THCus are generally recognised as safe (GRAS) in the USA since 2019. THCus can be used in foods and beverages in the USA with the 90th percentile all-user estimated intake of 239.20 mg/person per day.
While there is no history of use for the novel food, it is noted that the source of the novel food is widely consumed and over consumption of turmeric and curcuminoids is a potential health risk to consider in this assessment.
The whole organism from which the novel food is derived, and its naturally occurring metabolites, have been reviewed by the Committee on Toxicity of Chemicals in Food, Consumer Products and the Environment (COT) (COT, 2024). The Committee concluded that “substantial exceedances of the ADI (acceptable daily intake) [of curcuminoids] represents a potential health risk to humans, especially if other medicines are being taken concomitantly and for individuals with altered hepato-biliary function.” This may lead to rare but serious cases of idiosyncratic hepatotoxicity [IDH] in consumers.
THCus are synthetically derived and are not naturally occurring in the whole organism. However, THCus are produced from curcumin in vivo during metabolism in the liver through Phase I metabolism by hepatic reductases. The Committee also noted in their report that “synthetic forms of curcumin or chemical alterations are sometimes used in an effort to increase its bioavailability, thus potentially altering its toxicity profile.” Therefore, the ADI for THCus has been assessed for safety using data relevant to the novel food rather than curcumin and/or curcuminoids.
This information highlighted the question of exposure to curcuminoids from all sources in the diet, and this has been considered in this assessment of the novel food.
2.1.6. Proposed Use and Intake
The applicant intends to market the NF as a food supplement, as defined in The Food Supplements (England) Regulations (2003) and associated legislation in the nations of GB for those 18 years of age or over only, excluding pregnant and lactating women, at a maximum dose of 140 mg per day. The anticipated intake corresponds to the use of THCus in food supplements only, i.e. maximum 140 mg per day.
To address the combined exposure from the novel food and metabolism of curcuminoids in the diet, the applicant compared the estimated systemic exposure to THCus from the diet of 1.82 mg per day to the maximum oral intake of the NF of 140 mg per day. For systemic exposure to THCus from the diet, the applicant has assumed low oral bioavailability of approximately 1% and complete biotransformation of native curcumin to THCus. Therefore, based on these conservative assumptions, the estimated systemic exposure from the diet (1.82 mg per day) exceeds the systemic exposure from the NF (1.4 mg per day if 1% oral bioavailability is also assumed).
The applicant has therefore, not demonstrated that consumers would not be exposed to a high level of THCus from both the novel food and the background diet, given that THCus can be provided by the body from metabolism of other sources of curcuminoids. The potential combined exposure has been considered in this assessment but could not be quantified. Risk managers are advised to consider the need for information to consumers to minimise the potential for consumption of the novel food and other significant sources of curcuminoids in the diet; including turmeric and other turmeric or curcumoid supplements being consumed on the same day.
The following labelling (which is the same for the EU) was proposed by the applicant to restrict the intake of curcuminoids from other sources, : “This food supplement should not be consumed on the same day as other food supplements containing curcumin and/or curcuminoids” (European Commission, 2022). Risk managers may wish to consider if information to consumers is needed on the risk from exposure to curcuminoids from a range of sources in the diet.
This safety assessment took account of EFSA’s positive opinion on the novel food as part of the evidence to support the conclusions of the FSA and FSS. While EFSA reviewed a higher intake level of 300 mg per day, the maximum proposed intake level was derived as 140 mg per day, which is lower than the use level as proposed by the applicant to EFSA. The applicant has chosen to seek authorisation for a lower level of 140 mg per day in the UK. As such, the UK is reviewing a maximum intake of 140 mg per day only.
The novel food may contain palladium at < 5 mg/kg. At the proposed conditions of use, palladium intake from the novel food may thus reach up to 0.7 µg per day. This is well below the Permitted Daily Exposure (PDE) for palladium of 100 µg per day, as established by the Committee for Human Medicinal Products of the European Medicines Agency (EMA, 2022). There is no concern with respect to the exposure to contaminants from the consumption of novel food at the proposed uses.
2.1.7. Exposure Assessment
The 2024 COT report identified the potential for rare cases of hepatoxicity from curcuminoid supplements, especially their more bioavailable synthetic forms such as THCus (COT, 2024). As curcuminoids from the diet can be converted to THCus in the liver, an exposure assessment was requested from the applicant estimate potential combined exposure of THCus from conversion of curcuminoids in the diet, in addition to intake of the NF. The applicant performed an exposure assessment to evaluate the dietary exposure to THCus from both the NF and other food sources. In accordance with the target population for the NF, the exposure assessment was conducted for adults ≥18 years only, excluding pregnant and lactating women, and children and adolescents (<18 years). Combined intake with other curcumin and/or curcuminoid-containing supplements was not considered as the applicant’s proposed conditions of use for this NF recommends restricting the use of other curcumin and/or curcuminoids-containing supplements.
The applicant used the UK NDNS data to estimate consumption of ground turmeric in the target population. Resultant curcuminoid exposure was estimated by scaling for the average curcuminoid content of ground turmeric as 5.7% (Li et al., 2011). The proportion of turmeric consumers was similar between adult males and females, ranging from 6.0% to 7.0% of the surveyed population. Exposure estimates were calculated based on ‘consumers only’. Mean and 95th percentile exposures to curcuminoids were estimated and expressed in mg/kg bw per day (Table 4).
Mean exposure to curcuminoids was highest for females at 0.4 mg/kg bw per day, versus males at 0.3mg/kg bw per day. 95th percentile consumer-only exposure to curcuminoids were 1.1 mg/kg bw per day and 1.0 mg/kg bw per day in females and males respectively. This assessment only included exposures to curcuminoids from ground turmeric and did not include other dietary sources of curcuminoids (such as colouring agents).
An EFSA assessment of curcumin as a food additive estimated UK adult population exposure for curcumin from naturally occurring sources (e.g., turmeric used as a spice and in curry powder) and from its use as a food additive (colouring agent) (EFSA ANS Panel, 2010). The estimated mean curcumin dietary exposure in upper-limit (UL) adults was slightly higher than the NDNS calculation, at 1.0 mg/kg body weight per day, and the 95th percentile was estimated at 2.6 mg/kg body weight per day.
The applicant used the most conservative exposure value of 2.6 mg/kg body weight per day from the 95th percentile from the EFSA ANS Panel (2010) to calculate combined exposure of THCus from the NF and the diet.
Native curcumin exhibits very low oral bioavailability of approximately 1% of oral intake (Nelson et al., 2017; Yang et al., 2007). The applicant estimated that all bioavailable curcumin from naturally occurring dietary sources is converted to THCu following oral intake, which is a conservative estimate due to incomplete transformation and alternative metabolism of curcumin in the body. Using a reference body weight of 70 kg for adults, this corresponds to an estimated systemic THCu exposure from the diet in UK adults of approximately 1.82 mg per day at the 95th percentile intake level.
It is noted that the novel food is intended as an alternative source of curminoids, but combined exposure from different sources has been considered. The combined exposure of THCu of an estimated 1.82 mg per day (systemic) in the diet and from the use of the NF may increase THCu levels beyond the intended 140 mg per day (oral) established safe level, and proposed by the NF supplement.
The combined exposure of THCus is above the ADI for curcumin of 3 mg/kg bw (approximately 210 mg per day for a 70kg adult) established by JECFA in 2004 and confirmed in the evaluation by EFSA in 2010 (EFSA ANS Panel, 2010; World Health Organization, 2017). However, it is not appropriate to compare THCu exposure to the ADI for dietary curcumin as synthetic forms could have altered TK profiles and increased bioavailability (COT, 2024). Therefore, the safe level of 140mg per person per day established for THCus using the available toxicological information (see 2.1.9 Toxicological Information) on THCus specifically is considered more appropriate for the purpose of this assessment. The dietary exposure of THCus could increase the consumer exposure to THCus and may lead consumers to exceed the 140mg NOAEL if using both the novel food and other significant sources of curcuminoids are consumed in the diet. As such risk managers may wish to consider consumer information so that consumers don’t consume the NF and other significant dietary sources of curcuminoids or turmeric on the same day.
EFSA concluded that the proposed intake of 140 mg per day for the NF does not raise safety concerns (EFSA, 2021). Upon review, the FSA and FSS determined that the NF is safe for the majority of consumers, given the proposed use to limit concurrent use of other food supplements containing curcumin and/or curcuminoids alongside the NF. However, it should be noted that in rare cases, the NF may lead idiosyncratic hepatotoxicity [IDH]. Therefore, it is recommended that risk managers consider whether there should be measures taken to restrict use by consumers with particular risks, such as altered liver function or those taking certain medication (COT, 2024). Consideration may also be required for restricting the use of substances associated with increases in bioavailability of curcuminoids (such as piperine) alongside intake of this novel food.
2.1.8. Absorption, Distribution, Metabolism and Excretion (ADME)
One animal study was provided by the applicant assessing the pharmacokinetic parameter of the novel food (Novaes et al., 2017). The novel food was dissolved in a mixture of 2% Dimethyl sulfoxide (DMSO) and 98% Polyethylene glycol (PEG)-400, and administered by gavage to male Sprague–Dawley rats (n = 3) at 500 mg/kg. Blood samples were collected at 0, 15 and 30 min, and 1, 2, 4, 6, 12, 24, 48 and 72 h post-dose. Urine samples were collected at 0, 2, 6, 12, 24, 48 and 72 h post-dose. Serum and urine samples were treated with b-glucuronidase to release any glucuronide conjugates.
A maximum serum concentration (Cmax) average of tetrahydrocurcumin of 6.8 µg/mL was achieved after 1 hour, indicating rapid absorption. The total amount of tetrahydrocurcumin excreted in urine was 8 µg at 24 h, indicating partial oral absorption, and partial elimination through the renal route.
Literature from the applicant provided evidence of excretion of tetrahydrocurcumin primarily via the biliary system (Heger et al., 2014; Holder et al., 1978; Jäger et al., 2014). Tetrahydrocurcumin is also produced from curcumin in vivo during metabolism in the liver. The EFSA assessment did not raise any safety concerns regarding the ADME of the novel food. In reviewing the assessment the FSA/FSS note the wider work on exposure to curcuminoids and the ADME of turmeric, and have taken this into account in reaching a conclusion on the safety of the novel food.
2.1.9. Nutritional Information
The nutritional profile for five batches of THCus showed no protein or carbohydrate (including fibre or starch), though the limits of detection were high: 0.27 and 2.7 g/kg respectively. Small quantities of fats (0.22-0.29%), sodium (0.001-0.002%), ash (0.03-0.05%) and moisture (0.19-0.35%) were reported. FSA and FSS agree with the EFSA assessment that the novel food is not nutritionally disadvantageous.
2.1.10. Toxicological Information
Six unpublished toxicological studies were performed using THCus to support the safety of the novel food.
2.1.11. Genotoxicity
Two bacterial reverse mutation tests were submitted, including one requested by EFSA due to use of a non-authorised solvent in production of the novel food in the initial test. The second assay was conducted using Salmonella typhimurium strains TA97a, TA98, TA100, TA1535, and TA102 by the Indian Institute of Toxicology according to OECD 471 guidelines (Indian Institute of Toxicology, 2004; OECD, 2020). The concentrations tested were 61.72, 185.18, 555.55, 1666.67 and 5000 µg/plate using DMSO as vehicle. The study was conducted with and without metabolic activation using S9 fraction. No changes to the number of revertant colonies compared to controls were observed for all doses. The results demonstrate that THCus did not induce mutation in S. typhimurium under the conditions of this study.
An in vitro mammalian erythrocyte micronucleus test was conducted according to OECD test guideline 487 (Bioneeds India Private Limited, 2019; OECD, 2023). A trial test to assess cytotoxicity of THCus used concentrations of 0.0625, 0.125, 0.25, 0.5 and 1 mg/mL. Excessive cytotoxicity was observed for 0.5 and 1 mg/mL (cytotoxicity of 84.8% and 96.7% respectively), and as such these were excluded from the micronucleus experiment.
For the micronucleus test, THCus were assessed in proliferated lymphocytes in duplicates by exposing for short term (3 or 6 hours, without and with S9 mix for metabolic activation) and long-term treatment (20 to 24 hours, without metabolic). No statistically significant increase in the percentage of micronuclei in binucleated cells observed in any of the tested concentrations. As such the novel food was not considered aneugenic or clastogenic.
EFSA concluded that there are no concerns regarding the genotoxicity of the novel food, as demonstrated by the results of the two bacterial reverse mutation test and the in vitro micronucleus test. Upon review, the FSA and FSS agree with this decision and consider the novel food is not genotoxic.
2.1.12. Sub-acute toxicity
A sub-acute 14-day single dose oral toxicity study was conducted in rats following ISO 9002 guidelines (Indian Institute of Toxicology, 2004; International Organisation for Standardisation, 2016). Five male and five female Sprague–Dawley rats received one 2,000 mg/kg body weight (bw) dose of the novel food by gavage. All animals survived the 14-day observation period. The LD50 was set at 2000 mg/kg body weight per day. The FSA and FSS do not recommend the use of acute animal studies in the assessment of foods.
2.1.13. Sub-chronic toxicity
A 90-day repeated dose oral toxicity study was performed using the novel food in compliance with OECD guidelines 408 (Majeed et al., 2019; OECD, 2018). After five days of acclimatisation, adult Wistar rats (ten rats/sex assigned to the main study group, and five rats/sex assigned for recovery study group) were administered one 10 mL/kg dose per day for 90 days with the vehicle (0.5% carboxy methyl cellulose; CMC) or test item formulation (100, 200, or 400 mg/kg bw per day) via gavage.
There were no significant test item related findings in the gross pathology or histopathology. Statistically significant differences were observed as follows:
Cholesterol levels were significantly increased in the male group, and triglycerides were significantly decreased in the female group when treated with 400 mg/kg body weight per day. Animals recovered and there was no significant difference in these parameters for the respective recovery group. A lower blood platelet count was seen in males in the 400 mg/kg body weight per day treatment group. Lymphocytes and monocytes were higher in females in the 200 and 400 mg/kg body weight per day group, while in males, monocytes were lower in the 200 and 400 mg/kg body weight per day treatment groups.
The FSA and FSS agree with the EFSA panel that the observed differences were small, partly without a clear dose–response relationship, and that they reflect normal biological variation rather than adverse effects. The no observed adverse effect level NOAEL of this study is the highest dose tested, i.e. 400 mg/kg body weight per day. Once an uncertainty factor of 200 is applied (10 (interspecies variability) x 10 (intraspecies variability) x 2 (subchronic to chronic study duration)), the EFSA Panel derived a safe level of 2 mg/kg body weight or 140mg for a 70kg adult. The intended and expected maximum intake for the novel food is 140 mg per day, and as such the observed effects were not considered to present a hazard.
2.1.14. Reproductive and developmental toxicity
A reproductive and developmental toxicity study was performed using the novel food in compliance with OECD guidelines 421 (Majeed et al., 2019; OECD, 2016). The novel food or vehicle control (0.5% CMC) was administered via gavage to adult Wistar rats (10/group per sex) for 90 days at doses of either 0, 100, 200 or 400 mg/kg body weight per day.
During gestation maternal body weight in the 400 mg/kg body weight per day group was statistically significantly lower (by 6.6–8.6%) than in the control group. There were also some statistically significant differences in food consumption (both increased and decreased) between groups during some weeks.
Even though reported as not statistically significant, the EFSA panel noted parameters indicating adverse effects of the novel food in the 400 mg/kg body weight per day group. These were: higher mean post-implantation loss (difference between implantations and live foetuses/pups in percent) of 30.0% in the 400 mg/kg body weight per day as compared to 15.2%, 12.9% and 16.7% in 0, 100 and 200 mg/kg body weight per day groups, respectively. A lower live birth index of 83.1% was also reported in the 400 mg/kg body weight per day as compared to 95.8%, 100% and 98.9% in 0, 100 and 200 mg/kg body weight per day groups, respectively.
The NOAEL of this study is 200 mg/kg body weight per day. Once the standard uncertainty factors of 100 (10 (interspecies variability) x 10 (intraspecies variability)) is applied, the safe level is calculated as 2 mg/kg bw per day, or 140mg for a 70kg adult. The FSA and FSS also recognise that for GB the applicant has adjusted the maximum intended intake to 140 mg/kg body weight per day accordingly after evaluation by the EFSA panel.
2.1.15. Human studies
An uncontrolled study was conducted in 20 human adult subjects that received 300 mg of the novel food orally once per day for 28 days (Sabinsa Japan Corporation, 2011). The parameters measured at the end of the study were as follows: blood pressure, body weight/BMI, haematology, clinical chemistry and recording of adverse events. No statistically significant changes were found compared to baseline, and no adverse events were reported during the study.
The FSA and FSS agree with the EFSA panel that this uncontrolled study is of little value for the safety assessment of the novel food.
2.1.16. Incidence of idiosyncratic hepatotoxicity
Idiosyncratic hepatotoxicity (IDH) represents a rare and unpredictable form of liver injury that occurs independently of both the administered dose and the duration of exposure. It can occur in 1/500 to 1/50,000 individuals exposed to a particular drug, and the prevalence of idiopathic hepatitis in the community is estimated to be 1/100,000 (Kaplowitz, 2005).The potential of IDH from curcuminoids was evaluated by the COT and was identified as a potential safety concern for curcuminoid-containing supplements. Individuals prone to this response would likely not know they are susceptible before taking a supplement. Therefore, the applicant was requested to consider the potential for IDH due consumption of the NF.
The applicant notes that as idiosyncratic hepatotoxicity is unpredictable and dose-independent, it is not possible to exclude totally the risk of IDH from the NF. The applicant provided nutrivigilance data on the NF, reporting approximately 24.2 million doses (of 140 mg each) sold in Europe since 2022. No data or complaints regarding idiosyncratic hepatoxicity have been reported to the applicant. The applicant also noted that no animal models are available to reproduce the phenomenon of transient hepatotoxicity that resolves with continued drug exposure via physiological adaptation. In addition, due to the rare and unpredictable nature of the response, the sample size required for assessment in a human trial would be approximately 30,000 subjects and would not be feasible.
As idiosyncratic hepatotoxicity is a rare, unpredictable, and dose-independent condition, consumers may not necessarily be aware of their potential risk. Risk management may wish to consider if additional labelling may be appropriate to inform consumers of this rare risk.
2.1.17. Allergenicity
Protein was not detected in the novel food up to the limit of detection (0.27 mg/g). Therefore, the FSA and FSS agree with the EFSA Panel that the risk of allergic reactions to the novel food is low.
3. Other Regulators Opinions and Conclusions
THCus are manufactured by hydrogenating the curcuminoids from the rhizomes of the Curcuma longa (turmeric) plant, then dried to yield the purified powder. The turmeric rhizome and extract of the rhizome containing curcuminoids are not classified as a novel food when used in food supplements, and as such are permitted for sale in the EU and UK. THCus are obtained by hydrogenating the curcuminoids extracted from turmeric, and as such are considered as a novel food and should therefore be authorized before any consumption in GB.
The FSA/FSS considered that the provided toxicology studies did not raise any safety concerns under the proposed conditions of use. A review by the Committee on Toxicity of Chemicals in Food, Consumer Products and the Environment concluded that substantial exceedances of the ADI [of turmeric] represents a potential health risk to humans (COT, 2024). Therefore, risk management may wish to consider restriction on the use of compounds or delivery method in THCus food supplements which may increase bioavailability, for example piperine, similar curcumin compounds, or novel delivery methods.
The FSA/FSS also considered that there is a rare risk for idiosyncratic hepatotoxicity in a small portion of the population due to intake of the NF. The possibility of this unexpected idiosyncratic response may wish to be considered by risk managers when providing guidance on the use of such supplements.
In their application to the EFSA the applicant proposed an intended use of 400 mg/kg body weight per day, and the EFSA panel decided upon a maximum intake of 140 mg/kg body weight per day based on the results of the toxicological study. Even though reported as not statistically significant, the EFSA Panel noted adverse effects in the high-dose group (400 mg/kg body weight per day) during the reproduction/developmental toxicity study were observed, and consequently the NOAEL of the study was determined to be 200 mg/kg body weight per day. Once the standard uncertainty factors were applied to each NOAEL the safe level of 2 mg/kg body weight per day was identified. This corresponds to 140 mg of the novel food per day for the target population with a default body weight of 70 kg. This is in correspondence with the use level as proposed by the applicant.
In agreement with the EFSA safety assessment, consumption of the novel food under the proposed conditions of use (140 mg of the novel food per day for adults excluding pregnant and lactating women) is not expected to be a safety concern for the majority of the population. However, additional consideration was given to the potential for idiosyncratic hepatotoxicity in a small percentage of consumers, even at levels below the ADI, which may need risk management consideration (COT, 2024).
Allergenicity was not considered a risk due to no detection of protein in the novel food.
4. Uncertainties and limitations
The FSA and FSS noted that no specific uncertainties were flagged in the assessment.
Given the novel food is in the form of a supplement, there is little uncertainty around the anticipated intake and overall exposure from combined intakes given THCus are not known to be found in other foodstuffs. THCus are produced from curcumin in vivo during metabolism in the liver through Phase I metabolism by hepatic reductases. Therefore, significant consumption of turmeric or curcumin-containing supplements may raise the quantity of THCus in the body. Risk management may wish to consider restriction on the use of other curcumin compounds or sources alongside intake of the novel food. Idiosyncratic hepatotoxicity is also an unpredictable risk which may need management.
The FSA and FSS could not have completed the assessment of the novel food under the proposed conditions of use without the following data claimed as proprietary by the applicant:
-
Identity of the novel food as confirmed by HPLC.
-
Method validation reports for the chemical determination of THCus, heavy metals, solvents, and sodium in the novel food.
-
Toxicological information, including in vitro genotoxicity studies, subacute and sub-chronic toxicity studies, and a test item human study.
5. FSA & FSS conclusions for GB assessment
The application has been assessed/evaluated in line with 'Guidance on the preparation and presentation of an application for authorisation of a novel food in the context of assimilated Regulation (EU) 2015/2283 (EFSA NDA Panel, 2016), and assimilated Commission Implementing Regulation (EU) 2017/2469, for purposes of the GB safety assessment.
The conclusions of the EFSA opinion (EFSA NDA Panel, 2021), which have been reviewed in detail by the FSA and FSS for the purposes of the GB assessment, are considered appropriate and consistent within the identified caveats and uncertainties (uncertainties and limitations – see heading in section 5) identified (by EFSA or in the EFSA opinion).
6. Outcome of the assessment
The FSA and FSS have reviewed the applicant’s dossier, supporting documentation, and most notably the EFSA opinion, and consider that there is sufficient evidence to conclude the safety assessment of tetrahydrocurcuminoids (THCus). Further information was requested from the applicant on combined dietary exposure of THCus an additional toxicological considerations.
The FSA and FSS conclude that THCus is safe under the proposed conditions of use. The anticipated intake levels and the proposed use in food and food supplements is not considered to be nutritionally disadvantageous.
In making this assessment, the FSA and FSS were able to rely on sufficient scientific evidence to make a conclusion on safety with no further questions to the applicant, and therefore no further risk assessment activities are necessary.
Sufficient evidence was available in the literature to give the FSA and FSS confidence about the safety of this novel food, for example, where other national food safety authorities had positively assessed the application using the same risk assessment guidance and core legal requirements which apply in GB.
The Applicants provided sufficient relevant information as requested by FSA and FSS. The FSA and FSS review did not find any issues of divergence from the EFSA guidance or mutual approaches or new scientific issues for consideration.
There were no other specific issues that would require an assessment for the UK or the nations of the UK. In reaching this conclusion the work of the Committee on Toxicity of Chemicals in Food, Consumer Products and the Environment on turmeric and its natural metabolites (COT, 2024) were taken into account.
The FSA and FSS agree with the conclusions of the EFSA Panel that the novel food, tetrahydrocurcuminoids from turmeric (C. longa L.) is safe for the target population as a food supplement at 140 mg per day excluding children and pregnant and lactating women. However, for some unidentified consumers there may be a safety concern use to idiosyncratic hepatotoxicity. This may be managed with labelling to inform consumers of the potential risk.
Abbreviations
Updated 20/05/26: Correction to applicant’s name within the abstract.